Have you ever wondered why stainless steel, known for its rust resistance, sometimes still shows signs of rust? This article explores the science behind stainless steel’s composition and the factors affecting its durability. You’ll learn practical tips to prevent rust and maintain its shine.


Stainless Steel Definition
Stainless steel is a type of steel that contains less than 2% carbon (C) and more than 2% iron.
In the process of making stainless steel, alloying elements such as chromium (Cr), nickel (Ni), manganese (Mn), silicon (Si), titanium (Ti), and molybdenum (Mo) are added to improve its performance and make it resistant to corrosion, or rust-free. This is what gives the steel its name: “stainless.”
The process of making stainless steel involves adding different types and amounts of alloy elements to produce various levels of performance.
To distinguish between these different levels, stainless steel is categorized into different grades.
The table below lists the common alloy elements used in decorative stainless steel and is provided for reference purposes only.
Stainless Steel Chemical Composition Chart (percentage %)
| Steel Grades | C | Si | Mn | P | S | Cr | Ni |
|---|---|---|---|---|---|---|---|
| 304 | ≤0.08 | ≤1.00 | ≤2.00 | ≤0.045 | ≤0.03 | 18-20 | 8-10 |
| 301 | ≤0.15 | ≤1.00 | ≤2.00 | ≤0.045 | ≤0.03 | 16-18 | 6-8 |
| 202 | ≤0.15 | ≤1.00 | 7.5-10 | ≤0.05 | ≤0.03 | 17-19 | 4-6 |
| 201 | ≤0.15 | ≤1.00 | 5.5-7.5 | ≤0.05 | ≤0.03 | 16-18 | 3.5-5.5 |

Alloy Element Content
Generally, if the chromium content is 10.5%, the stainless steel is less likely to rust.
The higher the chromium-nickel content, the greater the resistance to corrosion.
For example, 304 stainless steel with a nickel content of 8-10% and a chromium content of 18-20% will typically not rust.
The manufacturing process of stainless steel can impact its resistance to corrosion.
Large stainless steel factories with advanced smelting technology, equipment, and techniques have better control over alloying elements, impurity removal, and billet cooling temperatures, resulting in more stable and reliable product quality. As a result, the stainless steel produced by these factories is less likely to rust.
In contrast, small steel plants with poor equipment and technology often have difficulty removing impurities during the smelting process, causing their stainless steel to be more prone to rusting.
External Environment
Stainless steel in a dry, well-ventilated environment is less likely to rust.
However, in high humidity, continuous rainy conditions, or environments with high acidity or alkalinity, stainless steel is more susceptible to rusting.
Even 304 grade stainless steel can rust if the surrounding environment is poor enough.

Many customers go to the market with a small magnet in their pocket to test if the stainless steel is magnetic, believing that non-magnetic stainless steel is rust-free.
However, this is a common misunderstanding. The magnetic property of stainless steel is determined by its structure, not its rust resistance.
Different solidification temperatures during the manufacturing process result in different structures of stainless steel, such as ferrite, austenite, and martensitic. Ferrite and martensitic stainless steel are magnetic, while austenitic stainless steel has good mechanical properties and weldability but is not as corrosion-resistant as ferrite stainless steel.
The market also circulates low-nickel, high-manganese content stainless steel in the 200 and 300 series, which are non-magnetic but have lower performance compared to 304 stainless steel with a high nickel content.
Additionally, 304 stainless steel may exhibit slight magnetic properties after undergoing drawing, annealing, polishing, and casting processes.
Therefore, using magnetic properties to judge the quality of stainless steel is not a scientific method and can lead to misunderstandings.
Many customers go to the market with a small magnet in their pocket to determine if the stainless steel is magnetic, assuming that non-magnetic stainless steel is rust-free.
However, this is a false understanding. The magnetic property of stainless steel is determined by its structure, not its resistance to rust.
When customers see brown rust spots on the surface of stainless steel, they are often surprised, thinking that “stainless steel does not rust, if it does, then the steel quality must have a problem.”
In reality, this is a one-sided and misinformed view of stainless steel.
Stainless steel can still rust under certain conditions. It has the ability to resist atmospheric oxidation, but its resistance to corrosion in acid, alkali, and salt environments can change based on its chemical composition, state of interaction, conditions of use, and type of environmental media.
For example, 304 stainless steel has excellent anti-corrosion properties in a dry, clean atmosphere, but if it is moved to a coastal area, it will quickly rust in the salt-rich sea fog.
Therefore, not all types of stainless steel can resist corrosion and rust at all times.
Stainless steel forms a very thin, fine, and stable layer of chromium oxide on its surface, which prevents oxygen from infiltrating and oxidizing, and resists corrosion. But if this protective film is continuously damaged, air, liquid, and oxygen atoms will continuously infiltrate and separate metallic iron atoms, causing the surface to rust continuously.
There are many forms of damage to the protective film on stainless steel surfaces, including:
The conditions mentioned above can damage and corrode the protective film on the stainless steel surface.
To maintain a bright, rust-free metal surface, we recommend:
Using pickling paste or spray to help remove rust and reform a chromium oxide film can restore the stainless steel’s corrosion resistance.
It is crucial to properly wash the stainless steel with water after pickling to remove any contaminants and acid residue.
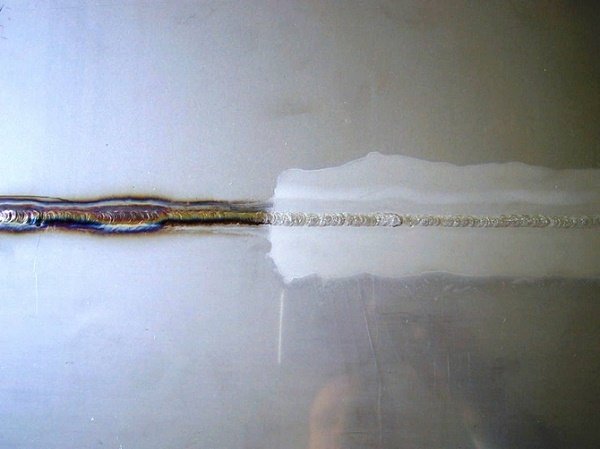
After all processes, use polishing equipment to repolish the stainless steel and seal it with polishing wax.
For small, mild rust stains, a mixture of 1:1 gasoline and engine oil can also be used to remove them.
Sandblast cleaning, cleaning with glass or ceramic particles, grinding, cleaning and polishing.

It is possible to erase previously applied materials, polishing materials, or cover polluted areas through mechanical means.
However, all forms of pollution, especially exotic iron particles, can be a source of corrosion, especially in humid environments.
Thus, it is best to clean the surface of the metal through mechanical means when it is dry.
Please note that mechanical cleaning can only clean the surface of the metal, not change its anti-corrosion ability.
We recommend using polishing equipment to repolish the surface after mechanical cleaning and sealing it with polishing wax.