Why is the meticulous process of metal surface pretreatment so crucial? In the realm of metalworking, achieving a pristine, smooth surface is more than aesthetic—it ensures durability and performance. This article delves into the intricacies of mechanical, chemical, and electrochemical methods to prepare metal surfaces for further processing. Readers will discover how these techniques enhance material properties, making metals ready for the next stage of manufacturing or finishing. Dive in to learn how pretreatment can make all the difference in metal quality and longevity.
“Surface pretreatment” refers to the mechanical, chemical, or electrochemical treatment of materials and their products before they undergo surface processing. This process is done to purify, roughen, or passivate the surface, making it ready for subsequent surface treatment or adjustment.
Metal surface pretreatment includes the following methods:
Surface leveling encompasses a variety of methods, including: mechanical polishing, chemical polishing, electrolytic polishing, rolling, brushing, sand blasting, and others.
The specific surface treatment process used depends on the condition of the parts and the technical requirements for the job.
The main objective of polishing is to make the rough and uneven surface of metal parts smooth and flat. Additionally, it can also remove burrs, scale, rust, sand holes, grooves, bubbles, and other surface imperfections on metal parts.
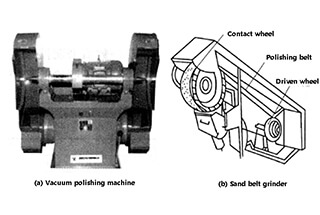
Polishing is performed using an elastic grinding wheel attached to a grinder. The working surface of the grinding wheel is covered with abrasive particles, which act like tiny cutting edges. When the grinding wheel rotates at high speed, the surface of the metal part is gently pressed against the working surface of the grinding wheel, causing the raised parts of the surface to be cut down and become smooth and flat.
Polishing can be used on all metal materials and its effectiveness depends on the characteristics of the abrasive, the rigidity of the grinding wheel, and the rotation speed of the grinding wheel. The abrasives commonly used for polishing include artificial corundum and emery. Artificial corundum, which is made up of 90-95% alumina and has a certain level of toughness, is widely used due to its less brittleness and the more edges and corners of its particles.
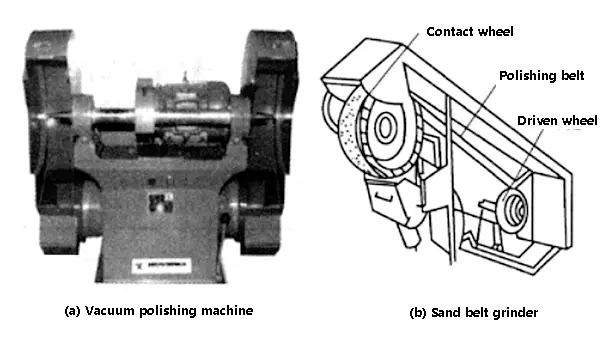
Fig. 1 polishing machine
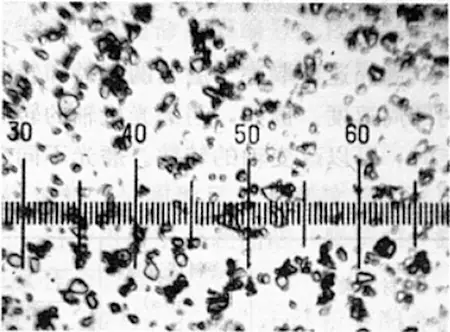
Fig. 2 Al2O3 abrasive (400X)
Abrasives can be classified into different grades based on particle size. The particle size of abrasives is usually determined by the number of holes per unit area (square centimeter) in a sieve. The higher the number of sieves, the smaller the holes. The particle size is represented by the number of abrasives that can pass through the sieve. The more abrasives that pass through, the finer the particle size, and the fewer abrasives, the coarser the particle size.
Table 1 highlights the characteristics and uses of common abrasives. Table 2 lists the most appropriate grinding wheel speeds for polishing different metal materials.
Table 1 Characteristics and uses of common abrasives
| Abrasive name | Artificial emery (SiC) | Artificial corundum (A2O3) | Natural emery | Silica sand (SiO2) |
| Mineral hardness / Mohs hardness | 9.2 | 9 | 7~8 | 7 |
| Toughness | Fragile | Relatively tough | Tenacity | Tenacity |
| shape | Sharp | Rounder | cylinder | Rounder |
| Particle size / mm (mesh) | 0.045~0.800(24~320) | 0.053~0.800(24~280) | 0.063~0.800(24~240) | 0.045~0.800(24~320) |
| Appearance | Purple black shiny crystal | White to gray black grain | Grayish red to black sand | White to yellow sand |
| Purpose | It is mainly used to polish low-strength metals (such as brass, bronze, aluminum, etc.) and hard and brittle metals (such as cast iron, carbon tool steel, high-strength steel) | It is mainly used to polish high-strength metals with certain toughness (such as quenched steel, malleable cast iron and manganese green steel) | Used for polishing general metals | General purpose grinding and polishing materials, also used for sandblasting and rolling |
Table 2 optimum grinding wheel speed for polishing different metal materials
| Material type | Steel, nickel, chromium | Copper and copper alloys, silver and zinc | Aluminum and aluminum alloy, lead and tin | ||
| Abrasive linear velocity / (M/s) | 18~30 | 14~18 | 10~14 | ||
| Suitable speed / (R / min) | Grinding wheel diameter / mm | 200 | 2850 | 2400 | 1900 |
| 250 | 2300 | 1900 | 1530 | ||
| 300 | 1880 | 1500 | 1530 | ||
| 350 | 1620 | 1530 | 1090 | ||
| 400 | 1440 | 1190 | 960 | ||
2.1 Mechanical polishing
Polishing is a type of grinding process. Some believe that it works by “tearing” atoms from the surface layer of the workpiece, causing the lower layer to instantly become smooth due to the action of surface tension before solidifying. Others consider polishing to be a result of surface tension. During the polishing process, the heat generated by friction can soften or even melt the surface, making it more than just a simple mechanical polishing process.
During polishing, the metal surface layer is melted but solidifies rapidly into an amorphous state due to the high thermal conductivity of the substrate metal. Before solidifying, the surface becomes smooth as a result of the combined effects of surface tension and friction from the polishing agent.
Workpieces that require a high degree of finish should undergo fine polishing after the initial polishing process.
Mechanical polishing is performed by using a polishing agent on the polishing wheel of a polishing machine. The polishing agent can be a polishing paste or polishing fluid. The former is a mixture of polishing abrasive and an adhesive, such as stearic acid or paraffin. The latter is a mixture of abrasive and an oil or water emulsion.
As the polishing wheel rotates at high speed, it removes slight unevenness on the workpiece and gives it a mirror-like shine. Mechanical polishing is used not only for surface pretreatment before plating, but also for finishing the coating after plating to improve the surface finish.
It is important to note that mechanical polishing is different from polishing. During polishing, obvious metal chips are cut away, whereas mechanical polishing does not result in significant metal loss. The high temperature generated by the friction between the high-speed rotating polishing wheel and the workpiece causes plastic deformation of the metal surface, filling in slight unevenness.
Additionally, the repeated removal of the extremely thin oxide film or other compound film formed on the metal surface under the influence of the surrounding atmosphere results in a flat and glossy surface.
2.2 Chemical polishing
Chemical polishing is a type of controlled chemical corrosion. It is a metal processing method that involves etching the metal in a specific polishing solution, making the surface flat and bright by selectively dissolving the metal.
Compared to other polishing techniques, chemical polishing has the advantages of simple equipment, low cost, easy operation, high efficiency, and being unaffected by the shape and structure of the parts. Additionally, compared to electrolytic polishing, chemical polishing does not require a power source and can be used to process workpieces with complex shapes. However, the production efficiency is higher but the surface processing quality is lower than that of electrolytic polishing.
Chemical polishing is a type of electrochemical process similar to that of electrolytic polishing. The reaction of chemical polishing belongs to the etching of a micro battery in the electrochemical process. Therefore, the principle of chemical polishing is similar to that of electrolytic polishing.
During the chemical dissolution process, an oxide film is generated on the metal surface, which regulates the speed of diffusion in the continuous dissolution process. The convex parts of the surface dissolve faster due to the thin thickness of the oxide film, while the concave parts dissolve slower.
A passive oxide film and an oxide film are constantly formed on the surface of steel parts, with the former being stronger than the latter. Due to the micro-unevenness of the surface, the micro-convex parts dissolve first, at a faster rate than the concave parts. The dissolution of the film and the formation of the film occur simultaneously, but at different rates.
As a result, the surface roughness of the steel part is leveled, resulting in a smooth and bright surface. Chemical polishing can effectively remove the surface damage layer caused by mechanical polishing, as it has a strong dissolving effect on the surface.
2.3 Electrolytic polishing
Electropolishing involves placing the workpiece as the anode and conducting electrolysis in a specific solution. During the process, the micro-protruding parts of the workpiece surface have high current density and dissolve rapidly, while the current density at the micro-recesses is low, causing the dissolution to be slow. This results in a flat and bright surface.
Electropolishing is commonly used for decorative finishing of parts such as carbon steel, stainless steel, aluminum, and copper, as well as surface finishing of some tools or for creating highly reflective surfaces and metallographic samples.
Phosphoric acid chromic anhydride polishing solution is widely used for iron and steel materials and contains components such as phosphoric acid, sulfuric acid, chromic anhydride, and additives like corrosion inhibitors, brighteners, and thickeners. The cathode is typically made of lead and the power supply voltage can be 12V.
In recent years, with the growing demand for stainless steel products, the demand for electrolytic polishing solutions has also increased. To prevent environmental pollution from electrolytic polishing solutions containing phosphoric acid and chromic anhydride, China has made efforts to develop environmentally friendly stainless steel electrolytic polishing solutions and has achieved significant progress.
Table 3 presents the solution composition and processing conditions of various new stainless steel electrolytic polishing solutions. Formulas 1 and 2 in the table do not utilize chromic anhydride, which resolves the issue of wastewater discharge and provides a pollution-free and environmentally friendly electrochemical polishing agent.
Table 3 solution composition and process conditions of environmentally friendly stainless steel electropolishing solution
| Solution composition and process conditions | Recipe 1 | Recipe 2 | Recipe 3 |
| Phosphoric acid (H3PO4,85%) /%Sulfuric acid (H2SO4,98%) /%Nitric acid (HNO3) /% perchloric acid /%Glacial acetic acid water (H2O)additive | 40~50 15~20 allowance Proper dextrin | 20~30 20~30 allowance Appropriate amount of glycerol | 10~15 8~10 allowance Small amount of additives |
| Temperature / ℃Current density / (A / dm2)Time / min | 60~70 20~30 3~5 | 65~70 15~30 3~8 | High temperature 10~30 3~5 |
Compared to mechanical polishing, electropolishing levels the polished surface through electrochemical dissolution, leaving no deformation layer on the surface and avoiding the mixing of foreign substances. Additionally, the electrolysis process results in oxygen precipitation, forming an oxide film on the polished surface, which improves its corrosion resistance.
Electropolishing is also useful for parts with complex shapes, wires, thin plates, and small parts, which are difficult to polish mechanically. Besides leveling, electropolishing can also remove surface inclusions and reveal defects such as cracks, sand holes, and inclusions on the surface of parts.
Rolling is a common method used for surface preparation before plating or surface modification after plating for large quantities of small parts. Rolling polishing is a process where parts and abrasives are placed together in a drum machine or bell machine for rolling grinding to remove burrs, roughness, and rust from the surface of parts and achieve a smooth surface.
In addition to abrasives, chemical reagents such as acid or alkali are often added during the rolling process. Thus, the rolling process serves to remove burrs, roughness, and rust as the parts and abrasives roll together, as well as the role of chemical reagents.
Fig. 3 is the schematic diagram of the calender.

Fig. 3 Schematic diagram of calender
Rolling polishing can eliminate oil stains and oxide scale on the surface of parts and produce a shiny surface. It can partially or completely replace polishing and polishing, but it is only suitable for large quantities of parts with low surface roughness requirements.
Rolling can be divided into the dry method and the wet method. The dry method utilizes abrasives such as sand, emery, broken glass, and leather, while the wet method uses steel balls, crushed stones, sawdust, lye, tea powder, etc. as abrasives.
The rotating speed during rolling is dependent on the characteristics of the parts and the drum structure, typically ranging from 15 to 50 RPM. If the speed is too high, the centrifugal force will prevent the parts from rubbing against each other in the drum, reducing the effectiveness of rolling. On the other hand, if the speed is too low, the efficiency is low.
When there is a large amount of oil stain or rust on the surface of parts during rolling, degreasing and etching should be performed first. If there is a small amount of oil stain, a small amount of alkaline substances or emulsifiers such as sodium carbonate, soap, or soap pod powder can be added for rolling. Dilute sulfuric acid or hydrochloric acid can be added for parts with rusty surfaces. After the parts are rolled in the acid medium, the acid solution should be rinsed immediately.
Brushing is a surface processing technique that utilizes a brushing wheel made of materials such as metal wire, animal hair, or natural or synthetic fibers. This method is mainly used to remove surface contaminants such as oxidation, rust, welding slag, old paint, and other debris. Additionally, brushing is also used to remove burrs left on the edges of a workpiece after machining.
The most commonly used brush wheels are made of steel wire or brass wire. If the workpiece material is hard, a high-rigidity steel wire brush wheel should be used in conjunction with a high speed. Conversely, for softer materials, a brass wire brush wheel is recommended.
Brushing can be performed either mechanically or manually. Both methods typically employ the use of a wet technique, with water being the most commonly used brushing solution. In some cases, a solution of 3% to 5% (by mass) sodium carbonate or sodium phosphate may also be used when brushing steel materials.
Sand blasting is a process that uses compressed air to blast dry sand, such as quartz sand, steel sand, or alumina, onto the surface of metal workpieces to remove surface defects such as burrs, scales, rust, carbon deposits, welding slag, molding sand residue, salt residue, old paint films, and dirt.
This method is commonly used for cleaning the surface of workpieces, such as removing residual sand and high carbon layers on castings and eliminating rust and scale on welds of weldments.
Sand blasting and acid washing are both techniques used for rust removal. However, while acid washing can cause hydrogen to penetrate into the interior of steel parts, increasing internal stress and reducing plasticity, sand blasting does not result in hydrogen embrittlement.
After sand blasting, workpieces made of high carbon steel, high-strength steel, or materials such as brass, stainless steel, and aluminum, can have improved adhesion of coatings or oxide layers. Hard chromium-plated and coated workpieces are typically cleaned using sand blasting. Machine tool accessories and measuring tools are often polished with sand blasting prior to milky white chromium plating.
Sand blasting is an effective method for surface pretreatment. It can completely remove impurities such as oxide scale, rust, old paint films, and oil stains from metal surfaces, resulting in a uniform metal color and a uniform roughness on the surface. This roughness can improve the bonding force between the anti-corrosion coating and the base metal, and increase the metal’s corrosion resistance.
Sand blasting is commonly used in thermal spray coating and plastic roughening treatments. Other surface roughening techniques include threading, knurling, electric spark roughening, and more.
There are two types of sand blasting: dry blasting and wet blasting. Wet blasting uses abrasives mixed with water to form a mortar, and a corrosion inhibitor is typically added to the water to prevent metal rust. Dry blasting is efficient but results in a rough surface, generates a large amount of dust, and causes the abrasive to break down more easily. On the other hand, wet blasting has minimal environmental impact, can have a decorative and protective effect on the surface, and is often used for more precise processing.
Etching is a process used to remove rust, oxide scale (formed during casting, forging, rolling, and heat treatment), and other corrosion products from the surface of a workpiece. This is achieved through the use of acid solutions, which have a strong ability to dissolve metal oxides. As a result, etching is also referred to as pickling.
For some non-ferrous metals, alkali etching may be used. Removing a large amount of oxides and poor surface structure is known as strong etching, while removing a thin oxide film on the workpiece surface to prepare it for electroplating is referred to as weak etching.
Inorganic acids, such as sulfuric acid, hydrochloric acid, nitric acid, phosphoric acid, and hydrofluoric acid, are typically used for pickling steel. Organic acids, such as acetic acid, fatty acid, and citric acid, can also be used. The action of organic acids is mild, and the residual acid has no significant after-effects. Additionally, the surface of the workpiece is clean after treatment and is less likely to re-rust.
Organic acids, although having the advantage of not causing significant after-effects, have a high cost and low rust removal efficiency, so they are mostly used for cleaning rust scale inside power equipment containers and other components with special requirements.
Inorganic acids, on the other hand, have high rust removal efficiency, fast speed, a wide range of raw material sources, and a low cost. However, if the concentration of inorganic acids is not properly controlled, the metal may become “over corroded,” and the residual acid is highly corrosive. If the acid solution is not thoroughly cleaned, it will impact the coating effect.
To slow down the corrosion and hydrogen embrittlement of metals, a suitable amount of buffers, such as rutin, urotropine, and thiourea, should be added to the rust removal solution.
(1) Pickling principle
The purpose of acid in pickling is to dissolve and mechanically strip the oxides from the surface of the workpiece. Using sulfuric acid as an example, sulfuric acid reacts with iron oxides (FeO, Fe3O4) to form ferrous sulfate and ferric sulfate.
Sulfuric acid reacts with the matrix iron through the gaps in the oxide scale, causing the dissolution of iron and the release of hydrogen. The reaction between sulfuric acid and matrix iron speeds up the chemical dissolution rate by reducing the low-solubility iron sulfate to high-solubility ferrous sulfate. The hydrogen produced under the oxide scale also creates mechanical top cracking and stripping effects on the oxide scale, improving pickling efficiency.
However, the reaction between sulfuric acid and matrix iron may result in excessive corrosion of the matrix and changes to the size of the workpiece. These are the disadvantages of using sulfuric acid in the pickling process.
The evolution of hydrogen during the pickling process may also result in hydrogen permeation of the workpiece, causing hydrogen embrittlement.
Hydrochloric acid primarily dissolves oxides. It reacts with iron oxide to form ferrous chloride and ferric chloride, both of which have high solubility. As a result, the mechanical stripping effect of hydrochloric acid etching is less pronounced than that of sulfuric acid.
For loose oxide scale, hydrochloric acid etching is fast, and there is less matrix corrosion and hydrogen permeation. However, for tight oxide scale, a large amount of acid is consumed when using hydrochloric acid alone. A mixed acid solution of hydrochloric acid and sulfuric acid is often used to achieve the mechanical stripping effect of hydrogen.
Nitric acid is mainly used for the treatment of high-alloy steel, and it is often mixed with hydrochloric acid for the treatment of non-ferrous metals. Nitric acid has a strong ability to dissolve iron oxides, and the solubility of ferrous nitrate and ferric nitrate is high, with minimal hydrogen evolution reaction.
When used in stainless steel, nitric acid does not cause matrix corrosion due to its passivating properties. However, when used in carbon steel, the issue of matrix corrosion must be addressed.
Hydrofluoric acid is primarily utilized for removing substances that contain silicon, such as alloying elements in certain types of stainless steel and alloy steel, mixed welding slag in welds, and residual molding sand on castings.
The combination of hydrofluoric and nitric acids is often used to treat stainless steel, but hydrofluoric acid is extremely corrosive and must be handled with caution.
Nitric acid releases toxic nitrides and can be challenging to dispose of, so extra care should be taken to avoid harm to the human body.
Phosphoric acid has good solubility for iron oxide and is less damaging to metal because it forms a water-insoluble phosphate layer (phosphating film) on the metal surface, which helps prevent corrosion.
Additionally, it makes an excellent base layer prior to painting. It is commonly used for removing rust from precision parts, but the cost of phosphoric acid is relatively high.
When phosphoric acid is used for rust removal, the main function is to turn the oxide scale and rust into water-soluble Fe (H2PO4) 3 and water-insoluble FeHPO4 and Fe3 (PO4) 2.
The diffusion of hydrogen is a weak process.
When using phosphoric acid for pickling, the amount of hydrogen produced is approximately 1/10 to 1/5 of that produced through hydrochloric acid or sulfuric acid pickling. Additionally, the rate of hydrogen diffusion and penetration is half that of the latter two acids.
Stainless steel and alloy steel have a complex composition and a dense structure for their oxide scale, making it difficult to remove in the derusting solution for ordinary carbon steel. A mixture of acids is typically used for this purpose.
When pickling alloy steel that contains titanium, it is necessary to add hydrofluoric acid.
The thick and dense oxide scale formed from heat treatment can be “loosened” in a hot, concentrated alkali solution containing a strong oxidant, and then etched using a mixture of hydrochloric and nitric acids, or sulfuric and nitric acids.
(2) Pickling additive
It is crucial to use a corrosion inhibitor in the pickling solution. The general belief is that a corrosion inhibitor can form an adsorption film or an insoluble protective film on the surface of the base metal in an acid solution.
The formation of this film occurs through an electrochemical reaction when the metal iron comes into contact with the acid, which charges the metal surface. The corrosion inhibitor, being a polar molecule, is attracted to the metal surface and forms a protective film, thus preventing the continuous action of the acid on the iron and achieving the goal of inhibiting corrosion.
From an electrochemical perspective, the formed protective film not only significantly blocks the anodic polarization process, but also promotes cathodic polarization, inhibits the production of hydrogen, and slows down the corrosion process.
Oxide scale and rust do not adsorb the polar molecules of the corrosion inhibitor to form a film because they interact with the acid through ordinary chemical action and do not have any charge on their surfaces.
Therefore, adding a specified amount of corrosion inhibitor to the derusting solution does not affect its derusting efficiency.
To evaluate the effectiveness of various corrosion inhibitors, it is crucial to determine their corrosion inhibition efficiency.
The corrosion inhibition efficiency can be determined by comparing the weight loss [g / (m2 · h)] of a sample with and without the corrosion inhibitor in the same medium and under the same conditions.
The specified amount of different corrosion inhibitors used in various acid solutions varies.
As the temperature of the acid washing solution increases, the inhibition efficiency of the corrosion inhibitor will decrease or even completely fail.
Therefore, each corrosion inhibitor has a specific allowable operating temperature.
The wetting agents used in pickling solutions are mostly non-ionic and anionic surfactants, with cationic surfactants being rarely used. This is because non-ionic surfactants are stable in strong acid mediums, and the only acceptable anionic surfactant is of the sulfonic acid type.
The use of surfactants with wetting, penetration, emulsification, dispersion, solubilization, and decontamination properties can greatly improve the pickling process and shorten the pickling time.
In order to minimize corrosion loss of the matrix, reduce the impact of hydrogen permeation, decrease acid mist, and improve the working environment, it is advisable to add an efficient corrosion and fog inhibitor to the pickling solution.
However, it is important to note that the corrosion inhibitor may form a film on the surface of the workpiece, which must be cleaned thoroughly. Additionally, the corrosion inhibitor can reduce the mechanical stripping effect of the hydrogen evolution reaction.
(3) Selection of acid type, concentration and temperature for pickling
The method of cleaning the surface of a workpiece depends on the material of the workpiece, the presence of rust and oxide scale, and the desired level of surface cleaning quality.
For steel workpieces, sulfuric acid, hydrochloric acid, or a combination of the two is commonly used.
To dissolve silicon-containing compounds on the surface of castings, hydrofluoric acid is added to either sulfuric acid or hydrochloric acid.
The concentration of sulfuric acid is typically around 20%. At this concentration, the etching speed of the oxide scale is fast and the damage to the underlying material is minimal.
The concentration of hydrochloric acid is usually below 15% as it produces fumes when the concentration exceeds 20%.
As the concentration of hydrochloric acid increases, the pickling speed accelerates and the pickling time decreases.
Table 4 illustrates the relationship between pickling time and acid concentration for steel workpieces with the same degree of corrosion in hydrochloric acid and sulfuric acid.
Table 4 Relationship between hydrochloric acid concentration and sulfuric acid pickling time of iron and steel
| Hydrochloric acid content /% | 2 | 5 | 10 | 15 |
| Pickling time/min | 90 | 55 | 18 | 15 |
| Sulfuric acid content /% | 2 | 5 | 10 | 15 |
| Pickling time/min | 135 | 135 | 120 | 95 |
| Hydrochloric acid content /% | 20 | 25 | 30 | 40 |
| Pickling time/min | 10 | 9 | / | / |
| Sulfuric acid content /% | 20 | 25 | 30 | 40 |
| Pickling time / min | 80 | 65 | 75 | 95 |
As the temperature increases, the pickling speed also increases and the time required is shortened.
Table 5 displays the relationship between pickling time and temperature for steel workpieces with the same level of corrosion in hydrochloric acid and sulfuric acid.
Table 5 Relationship between pickling time and temperature
| Acid content /% | Sulfuric acid pickling time/min | Hydrochloric acid pickling time/min | ||||
| 18℃ | 40℃ | 60℃ | 18℃ | 40℃ | 60℃ | |
| 5 | 135 | 45 | 13 | 55 | 15 | 5 |
| 10 | 120 | 32 | 8 | 18 | 6 | 2 |
(4) Pickling process of iron and steel workpiece
Pickling and rust removal methods include immersion pickling, spray pickling, and acid paste rust removal.
After undergoing degreasing treatment, the metal that has been impregnated and pickled is placed in an acid tank.
Once the oxide scale and rust have been removed, the metal is rinsed with water and neutralized with an alkali to produce a surface suitable for painting.
Table 6 provides information on the strong etching process parameters for steel workpieces.
Table 6 strong etching process parameters of steel workpieces
| Project | Forgings and stamping parts | General steel parts | Casting | ||
| 1 | 2 | 1 | 2 | ||
| Concentrated sulfuric acid / (g / L.) hydrochloric acid / (g / L) hydrofluoric acid / (g / L) Rodin / (g / L) Urotropine / (g / L) | 200~250 2~3 | 150~200 1~3 | 150~200 1~3 | 80~150 | 100 10~20 |
| Temperature / ℃ Time/min | 40 ~ 60 until all is divided | 30 ~ 40 until all is divided | 1.5 | Until 40 ~ 50 is divided | 30 ~ 40 until all is divided |
Electrochemical etching involves using electrolysis to strip the surface of a workpiece, which serves as the anode or cathode, in an acid or alkali solution. The process can also be accelerated by stirring the solution, which generates hydrogen at the cathode and renews the etching solution on the workpiece surface.
Electrochemical rust removal can be classified into anodic etching and cathodic etching, depending on the polarity of the workpiece.
During anodic etching, the oxide scale is removed through a combination of chemical and electrochemical dissolution of the workpiece metal, and mechanical stripping of oxygen.
In cathodic etching, the oxide scale is removed primarily through the mechanical effect of the large amount of hydrogen generated, and the reduction effect of primary atomic hydrogen on the oxide.
Anodic etching results in large, few oxygen bubbles with limited mechanical stripping effect, but if it takes too long, it can cause excessive corrosion of the underlying metal.
On the other hand, cathodic etching minimizes metal corrosion, preserving the size of the workpiece, but may lead to hydrogen permeation and ash residue.
Anodic etching is slow and corrosive to the base metal, making it only suitable for workpieces with a thin oxide scale. However, it does not cause hydrogen embrittlement.
On the other hand, cathodic etching is fast and does not result in over-corrosion of the workpiece, making it suitable for workpieces with thick oxide skins. However, it has the disadvantage of hydrogen permeation.
Currently, most methods used in China are either anodic etching or a combination of cathodic and anodic etching. Electrochemical etching is used for both strong and weak etching.
Compared to chemical etching, electrochemical etching is more effective in quickly removing the oxide scale firmly bonded to the metal surface. It is also less affected by changes in acid concentration and has little impact on the underlying material.
This method is easy to operate and manage, but requires specialized equipment and requires more hanging operations. There is also a risk of uneven dissolution of the oxide scale.
The advantages of electrochemical etching include fast etching speed, low acid consumption, and little influence of the iron ion content in the solution on the etching ability.
However, this method requires power supply equipment and consumes power.
Workpieces with complex shapes are difficult to etch because of the poor dispersion ability.
When the oxide scale is thick and dense, it should be pre-treated with sulfuric acid chemical etching to loosen the oxide scale before undergoing electrochemical etching.
Organic solvent degreasing is a common method for removing grease from metal materials. It works by using the physical dissolution properties of organic solvents on both types of oils.
Gasoline and kerosene are commonly used solvents, but chlorobenzene and kerosene are more affordable and less toxic alternatives.
Organic solvent degreasing is characterized by its no-heat process, fast degreasing speed, and lack of corrosion to the metal surface. It is particularly suitable for removing mineral oils with high viscosity and high melting points, which are difficult to remove with alkaline solutions.
Therefore, it is an appropriate pretreatment for almost all surface treatment technologies, especially for parts with severe oil pollution or metal parts that are susceptible to corrosion from alkaline degreasing solutions.
However, this method is not comprehensive and chemical and electrochemical methods may be necessary to supplement the degreasing process. Additionally, most organic solvents are flammable and toxic, and the cost can be high.
It is important to prioritize safety, take precautions, and maintain good ventilation during operation.
At present, chemical degreasing using an alkaline solution is widely used in production.
While the oil removal time for this method is longer than that of organic solvents, it has the advantages of being non-toxic, non-flammable, requiring simple equipment, and being inexpensive and easy to operate, making it a reasonable choice for oil removal.
The core of this method is to remove oil through saponification and emulsification. The former removes animal and vegetable oils, while the latter removes mineral oils.
With proper selection of the process, removing the two types of grease is not difficult.
However, when there are high requirements for the binding strength of the coating, relying solely on an alkaline solution for chemical oil removal of plated parts may not be sufficient.
This is particularly true when the oil stain is primarily mineral oil, as it takes a long time to remove and may not be completely removed due to the limited emulsification effect of the alkaline oil removal solution.
In such cases, it is necessary to use electrochemical (electrolytic) oil removal with stronger emulsification to achieve satisfactory results.
Electrochemical oil removal, also known as electrolytic oil removal, is a process of removing oil by placing metal parts in an oil removal liquid and using the parts as either the anode or cathode while connected to a direct current.
The composition of the electrochemical degreasing solution is similar to that of chemical degreasing solutions.
A nickel plate or nickel-plated iron plate is commonly used as the counter electrode, which serves only as a conductor.
Production experience has shown that electrochemical oil removal is several times faster than chemical oil removal, and effectively removes oil pollution. This is due to the mechanism of electrochemical oil removal.
Ultrasonic cleaning uses a high-frequency oscillation signal that is converted into high-frequency mechanical oscillation by a transducer.
The ultrasonic wave can effectively propagate in different media, including gas, liquid, solid, solid solution, and it can transmit strong energy. The ultrasonic wave is transmitted into the cleaning liquid in the tank through the tank wall and causes the microbubbles in the liquid to vibrate due to reflection, interference, and resonance.
The ultrasonic waves create strong impacts and cavitation on the interface, which is the basis of ultrasonic cleaning. The effectiveness of ultrasonic cleaning depends on various factors, including the type of cleaning fluid, cleaning method, cleaning temperature and time, ultrasonic frequency, power density, and the complexity of the parts being cleaned.
Common liquids used for ultrasonic cleaning include organic solvents, alkaline solutions, and water-based cleaning solutions.
The most commonly used ultrasonic cleaning and degreasing device consists of an ultrasonic transducer, cleaning tank, and generator. It may also include additional components for cleaning fluid circulation, filtration, heating, and transportation.
Ultrasonic cleaning is a popular method because of its simplicity, fast cleaning speed, and good results.
Using a low-temperature, high-efficiency cleaning agent to remove oil stains on metal surfaces is not only highly effective, but also energy-efficient due to its low cleaning temperature.
Vacuum degreasing cleaning is a new and environmentally friendly cleaning technology. It uses hydrogen carbide cleaning agent, which has minimal impact on human health, is less irritating, and has no odor.
This technology provides the same level of cleaning as triethanolamine, and is even more effective than alkali liquor. Additionally, the cleaning agent can be recovered and regenerated.
The vacuum degreasing cleaning device is a closed system that is pollution-free, has a high safety factor, is highly productive, and allows for automatic loading and unloading of materials, making it convenient to operate.
In the future, vacuum degreasing technology, whether with or without liquid cleaning, is expected to be widely used.
When conducting surface non-destructive testing on important large components such as aircraft to detect fatigue cracks and hard damage, the surface coating (paint) must be removed first.
Traditional methods of removing the coating include chemical stripping or manual grinding with a grinding wheel, but both methods have drawbacks. Chemical stripping can corrode and damage the metal matrix, while grinding with a grinding wheel can easily damage the substrate and is low in efficiency.
Recently, a new paint removal process using plastic shot spraying has been developed and has shown good results. This process involves spraying granular plastic onto the surface of the workpiece at high speed through a spray gun powered by compressed air.
The paint layer is stripped by the sharp edges and corners of the plastic shot cutting and impacting the surface. This provides an efficient way to remove paint.
Plastic shot paint removal has several advantages, such as being non-damaging to the substrate or coating due to the plastic shot having a hardness that is higher than the paint layer but lower than the substrate or coating and anodized surface layer. This also provides a clean surface for the new paint layer, improving its adhesion. Additionally, the plastic pellets can be recycled and easily separated from the peeled paint layer.
Ultrasonic sand blasting is a process of coarsening the surface of a substrate by using compressed air to spray hard sand particles at high speed onto the surface, resulting in a mechanical scouring effect. The speed of ultrasonic sand blasting is 300 to 600 meters per second and is more efficient than traditional sand blasting, with a blasting efficiency that is three to five times greater.
It is commonly used in the surface pretreatment of large structural parts, such as cleaning the surface before applying surface coating to bridges, ships, boilers, and pipelines. Additionally, it is often used for surface coarsening before spraying parts or large equipment with high requirements for spraying effects and cleaning equipment surfaces with heavy natural pollution, such as paint, cement, and organic or inorganic scale.
The coarsening treatment increases the “anchor hook” effect between the coating and the substrate, reducing shrinkage stress of the coating and improving the bonding strength between the coating and the substrate.
The sand used for sand blasting should have high hardness, density, crushing resistance, and low dust content. The particle size should be determined based on the required surface roughness. Commonly used sand grains include corundum sand (alumina), silica sand, silicon carbide, and emery.
Supersonic surface shot peening is a process in which supersonic projectiles are sprayed onto the surface of the workpiece, causing plastic deformation on the surface and forming a strengthening layer of a certain thickness.