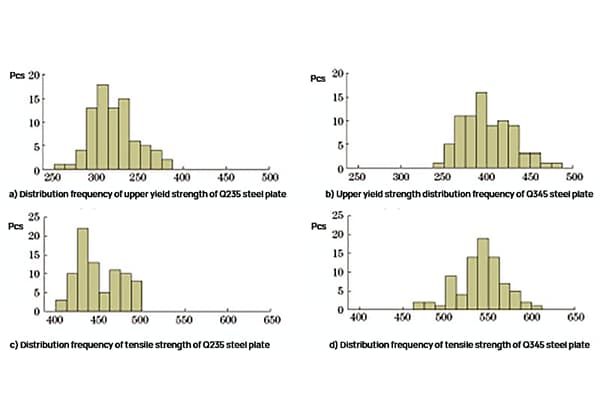
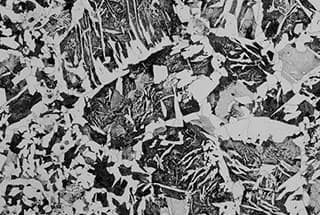
What makes some metals ideal for skyscrapers while others are perfect for electronics? The answer lies in understanding the key differences between ferrous and non-ferrous metals. This article dives into their unique properties, compositions, and varied applications, guiding you through what sets them apart and why it matters. By the end, you’ll grasp how these metals are pivotal in everything from construction to high-tech gadgets.

When it comes to metals, what comes to mind?

When it comes to metals, people often think of glittering jewelry or shining pots and pans. However, not many people associate metals with the color black. Yet, “black metal” is actually a large category of metals.
The term “black” can easily lead people to believe that ferrous metals must be black and nonferrous metals must be colorful. This belief leads many people to think that silver-white iron, chromium, and silver-gray manganese cannot be considered “black” metals.
However, iron, chromium, manganese, and their alloys are actually included in the category of ferrous metals. In the industry, these metals are collectively referred to as ferrous metals.
In reality, pure iron and chromium are silver-white, while manganese is silver-gray. The surface of steel is usually covered with a layer of black Fe3O4, which, along with the fact that manganese and chromium are mainly used to produce black alloy steel, can give the impression that these metals are “black.

Metals are a group of substances that are characterized by their luster, good conductivity, thermal conductivity, strong mechanical properties, and positive temperature coefficient of resistance. There are a total of 86 types of metals in the world.
People usually categorize metals into two groups: ferrous metals and nonferrous metals, based on their color, properties, and other distinctive characteristics.

Ferrous metals mainly consist of iron, chromium, manganese, and their alloys, such as steel, pig iron, ferroalloy, cast iron, etc. Steel and pig iron are iron-based alloys with carbon as the main addition element, and they are collectively referred to as iron-carbon alloys.
Pig iron is produced by smelting iron ore in a blast furnace and is mainly used for making steel and castings.
Cast iron, with a carbon content greater than 2.11%, can be obtained by melting pig iron in a furnace. The liquid cast iron can then be cast into castings, which are referred to as iron castings.
Ferroalloy is an alloy composed of iron and elements such as silicon, manganese, chromium, titanium, etc. It is one of the raw materials for steelmaking and serves as a deoxidizer and alloy element additive.
An iron-carbon alloy with a carbon content less than 2.11% is called steel, which is produced by melting pig iron used for steelmaking in a steelmaking furnace according to a certain process. Steel products include ingots, continuous casting billets, and direct casting into various steel castings.

Generally, the term “steel” refers to iron that has been rolled into various forms of steel. Despite the fact that there are only a few ferrous metal mines, each mine produces a large output, resulting in a total ferrous metal output that makes up 95% of the world’s metal output!
Iron, chromium, and manganese are the primary raw materials for smelting iron and steel, which play a crucial role in the national economy and serve as an important indicator of a country’s strength.
Related reading: What Is Ferrous Metal?
Nonferrous metals, also referred to as nonferrous metals in a narrow sense, include all metals except iron, manganese, and chromium. In a broader sense, nonferrous metals also encompass nonferrous alloys, such as copper, tin, lead, zinc, aluminum, brass, bronze, aluminum alloys, and bearing alloys.
A nonferrous alloy is an alloy made up of a nonferrous metal as the primary component (usually accounting for more than 50%) and one or multiple other elements. In industry, metals such as chromium, nickel, manganese, molybdenum, cobalt, vanadium, tungsten, and titanium are also used.
Related reading: 72 Different Types of Metals
These metals are mainly used as alloy additives to enhance the performance of other metals. Tungsten, titanium, molybdenum, and other metals are mainly used to manufacture hard alloys for cutting tools. These nonferrous metals are referred to as industrial metals.

Nonferrous metals can be divided into heavy metals (such as copper, lead, and zinc), light metals (such as aluminum and magnesium), precious metals (such as gold, silver, and platinum), and rare metals (such as tungsten, molybdenum, germanium, lithium, lanthanum, and uranium).
In 1958, China classified iron, chromium, and manganese as ferrous metals, and the remaining 64 metals as nonferrous metals. These 64 nonferrous metals include aluminum, magnesium, potassium, sodium, calcium, strontium, barium, copper, lead, zinc, tin, cobalt, nickel, antimony, mercury, cadmium, bismuth, gold, silver, platinum, ruthenium, rhodium, palladium, osmium, iridium, beryllium, lithium, rubidium, cesium, titanium, zirconium, hafnium, vanadium, niobium, tantalum, tungsten, molybdenum, gallium, indium, thallium, germanium, rhenium, lanthanum, cerium, praseodymium, neodymium, samarium, europium, gadolinium, terbium, dysprosium, holmium, erbium, thulium, ytterbium, lutetium, scandium, yttrium, and thorium.
Compared to pure metals, nonferrous alloys generally have higher strength and hardness, greater resistance, a smaller temperature coefficient of resistance, and good overall mechanical properties. Common nonferrous alloys include aluminum alloy, copper alloy, magnesium alloy, nickel alloy, tin alloy, tantalum alloy, titanium alloy, zinc alloy, molybdenum alloy, zirconium alloy, etc.

Due to the crucial role that rare metals play in modern industry, they are sometimes separated from nonferrous metals and classified into a separate category, resulting in three major categories of metals: ferrous metals, nonferrous metals, and rare metals.
Compared to ferrous metals, nonferrous metals have a much greater variety, and there are many more mining areas for nonferrous metals.
Today, nonferrous metals and their alloys have become essential structural and functional materials in various industries, including machinery manufacturing, construction, electronics, aerospace, and nuclear energy utilization.

In practical applications, nonferrous metals are generally divided into 5 categories:

At first glance, the distinction between ferrous and non-ferrous metals is straightforward. Ferrous metals contain iron, whereas non-ferrous metals do not.
However, there are more differences beyond this simple black-and-white definition. The composition and applications of ferrous and non-ferrous metals greatly vary.
Ferrous Metal Promised Durability:
Ferrous metals include iron and many forms of steel, which are widely used materials. Due to their durability, ferrous metals are commonly utilized for construction, piping, and the production of tools.
However, just because they are strong does not mean that they lack versatility. Steel, for instance, is a combination of iron and carbon, but by adjusting the ratio of these components or adding other materials, steel can be transformed into anything from sturdy steel beams used in high-rise buildings to gleaming stainless steel castings.

Examples of ferrous metals include:
Non ferrous metals guarantee flexibility:
Non-ferrous metals have a multitude of uses, with their versatility and unique properties making them ideal for a wide range of applications across various industries.
For instance, copper and aluminum have the ability to conduct heat and electricity, while many non-ferrous metals form the core components of most smartphones.
In addition, metals like gold and silver have been used for ornamental purposes for thousands of years.
One of the key advantages of non-ferrous metals is that they are inherently resistant to corrosion and magnetism, making them a popular choice for many applications.

Examples of non-ferrous metals include:
Main differences:
The primary differences between ferrous and non-ferrous metals are their cost and accessibility. Ferrous metals, such as carbon and iron, are abundant in their basic elements, making them easy to obtain and relatively inexpensive.
On the other hand, non-ferrous metals like gold and silver are scarce and, due to their widespread use, tend to be more expensive. This is why they are often referred to as “precious metals.”
Metals have played a crucial role in the development of human society, as they have been instrumental in many of the key transitions in human history. The Bronze Age and the Iron Age, which followed the Stone Age, are examples of how metal materials have shaped human society.
After World War II, the advancement of science and technology led to the development of new metal materials, such as ductile iron, alloy cast iron, alloy steel, heat-resistant steel, stainless steel, nickel alloy, titanium alloy, and cemented carbide, which have since been applied in practical production.
In modern times, the research and development of metal materials has expanded beyond the traditional realm of pure metal and pure alloys. With the advancement of basic theory and technology, new metal materials have emerged, including high-temperature structural materials such as nano metals, high/medium entropy alloys, directionally solidified columnar crystals, single crystal alloys, metal matrix composites, and shape memory alloys.
Metal materials have become an essential foundation for the development of human society. Agricultural modernization, industrial modernization, national defense, and scientific and technological progress are all supported by metal materials, from agricultural tools and mechanical parts, to sophisticated weapons like aircraft, missiles, fire arrows, satellites, nuclear submarines, as well as components or parts required for cutting-edge technologies like atomic energy, television, communications, radar, and electronic computers.
Currently, many countries around the world, especially those with developed industries, are competing to develop their metal and non-ferrous metal industries and increase their strategic reserves of metals.
In conclusion, metal materials are crucial basic materials and important strategic resources for the national economy, daily life, defense industry, and scientific and technological progress.