Heat treatment principle
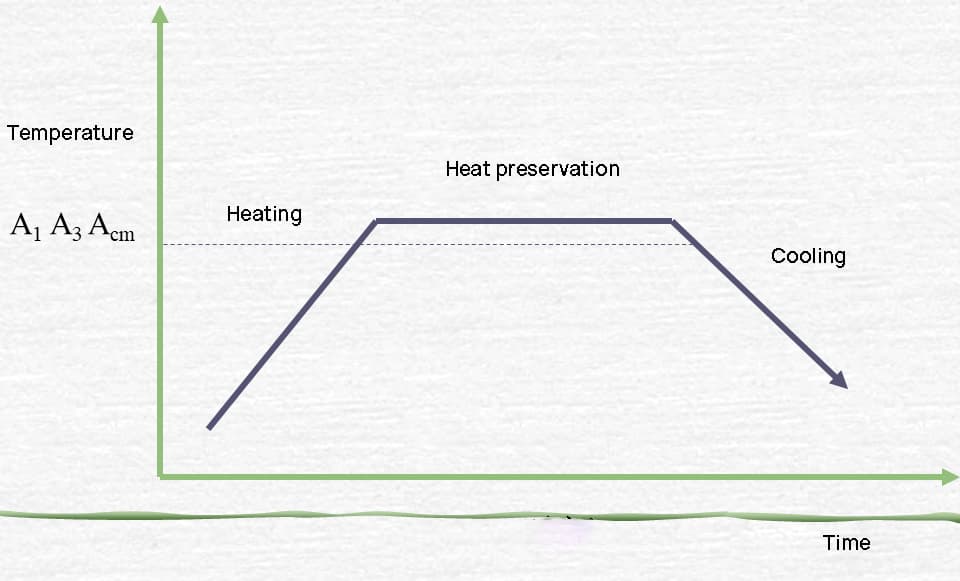
Transformation of steel during heating
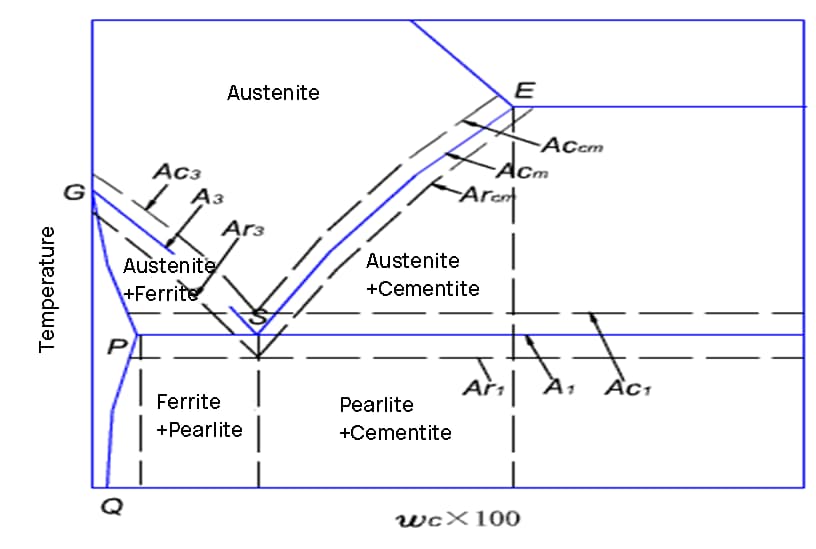
C-curve
The C-curve is a tool used for analyzing the transformation of carbon steel’s microstructure during cooling after it has been heated to form austenite.
There are two methods for cooling steel in the heat treatment process:
- Isothermal cooling transformation: This process involves cooling the steel, which has been heated to form austenite, to a specific temperature below the A1-line at a relatively fast rate, and then insulating it to allow the austenite to undergo structural transformation at a constant temperature.
- Continuous cooling transition: This refers to the continuous decrease in temperature that occurs over time during the cooling process.
In practical production, continuous cooling is the most commonly used method.
1. Cooling method of steel during heat treatment


Isothermal transformation curve of undercooled austenite in eutectoid steel

a. High temperature transition
The austenite of eutectoid steel is cooled to a temperature ranging from A1 to 550°C, resulting in a pearlite structure through the process of isothermal transformation. This transformation of austenite into pearlite is a result of the alternating nucleation and growth of ferrite and cementite, as depicted in Figure 3-7.
Formation of pearlite
Firstly, the nucleus of cementite crystals is formed on the grain boundary of austenite.
The carbon content of cementite is higher than that of austenite, leading to absorption of carbon atoms from the surrounding austenite.
As a result, the nearby austenite’s carbon content is reduced, creating conditions for the formation of ferrite and converting this part of austenite into ferrite.
The low carbon solubility of ferrite means that excess carbon must be transferred to adjacent austenite as it grows, causing the carbon content of the adjacent austenite region to increase and creating conditions for the formation of new cementite.
Through this process, austenite is eventually fully transformed into a pearlite structure with alternating layers of ferrite and cementite.

The formation of pearlite requires the movement of carbon atoms, with the distance of movement determining the width of the pearlite lamellae. At high temperatures, the movement of carbon atoms is more extensive, resulting in wider pearlite lamellae.
Conversely, at low temperatures, the carbon atoms have difficulty moving and therefore the pearlite lamellae are denser. The microstructure transformed from 727°C to 650°C is pearlite.

The structure obtained through transformation between 650°C and 600°C is known as sorbite, which is also referred to as fine pearlite. The transformation between 600°C and 550°C results in the formation of troostite, which is also known as very fine pearlite.

These three types of pearlite structures are only differentiated by their lamellar spacing and do not have any fundamental differences.
b. Intermediate temperature transition
The products of the isothermal transformation of austenite in eutectoid steel, from undercooling to a temperature range of 550°C to 240°C, belong to the bainite structure. The upper bainite is formed in the upper part of this temperature range, while lower bainite is obtained in the lower part. The lower bainite has improved hardness and strength, as well as better plasticity and toughness. However, the upper bainite does not have any practical applications.

c. Low temperature transition
It is highly challenging for carbon atoms in austenite to shift below 240°C.
Austenite only undergoes an isomorphic transformation, changing from a face-centered cubic (y-iron) structure to a body-centered cubic (α-iron) structure.
All carbon atoms within the original austenite stay in the body-centered cubic lattice, resulting in a supersaturated α-iron.
This supersaturated solid solution of carbon in α-iron is referred to as martensite.
Retained austenite
When eutectoid steel’s austenite is cooled down to 240°C (MS), it begins to transform into martensite.
As the temperature continues to drop, the amount of martensite increases while the undercooled austenite decreases.
By the time the temperature reaches -50°C (MF), the undercooled austenite has completely transformed into martensite.
Thus, the structure between MS and MF consists of martensite and retained austenite.
Due to variations in carbon content, martensite has two forms.
Martensite with a high carbon content takes on a needle-like shape, known as needle-like martensite.
Martensite with a low carbon content, on the other hand, is plate-like and referred to as plate-like martensite.

| Tissue | Carbon content (%) | Mechanical properties | |||
| HRC | (Mpa) | ak J/cm2 | Ψ(%) | ||
| Low carbon | 0.2 | 40~45 | 1500 | 60 | 20~30 |
| High carbon | 1.2 | 60~65 | 500 | 5 | 2~4 |
Table 4-5 comparison of properties of low carbon martensite 15MnVB steel and quenched and tempered 40Cr steel
| Steel grade | 15MnVB40Cr |
| State | Quenching and tempering state of low carbon martensite |
| HRC | 4338 |
| σo.2/MPa | 1133800 |
| σb/MPa | 13531000 |
| δ5(%) | 12.69 |
| φ(%) | 5145 |
| ak/Jcm-2 | 9560 |
| ak(-50℃)/J.cm-2 | 70≤40 |
(2) Continuous cooling
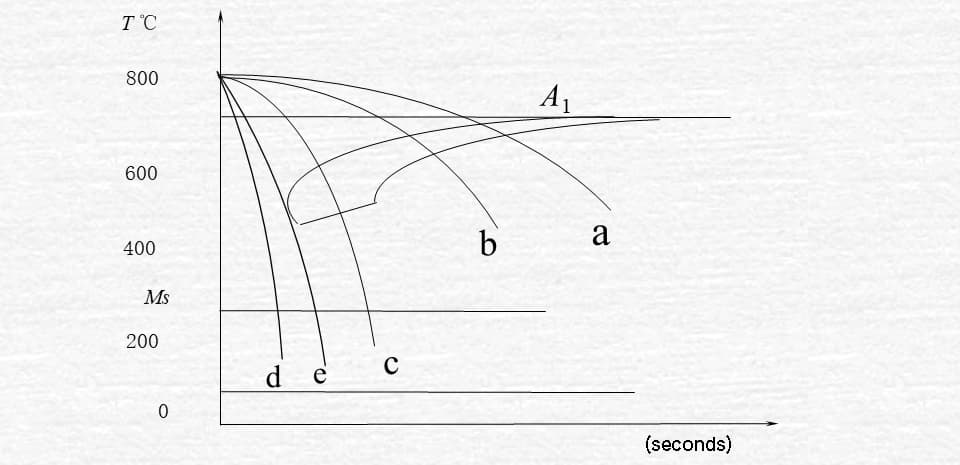
Fig. 3-9 cooling transformation curve of eutectoid steel
a. Cooling with furnace
When the cooling curve intersects with the start line for the pearlite transformation, the transformation of austenite to pearlite begins.
Once the cooling curve intersects with the end line of the transition, the transformation is complete.
As a result of the transformation occurring within the pearlite region, a pearlite structure is formed.
b. Cooling in air
As a result of the rapid cooling rate, the transformation takes place in the sorbite region, producing ferrite as the transformation product.
c. oil cooling
The cooling curve only intersects with the start line for the pearlite transformation (in the troostite transformation zone), but does not intersect with the end line.
As a result, only a portion of the austenite is transformed, resulting in the formation of troostite as the transformation product. The remaining portion of austenite transforms into martensite upon cooling to the MS line.
Finally, a mixed structure of martensite and troostite is obtained.
This refers to the product that has been cooled in oil.
d. Water cooling.
Due to the rapid cooling rate, the cooling curve does not intersect with the start line for the pearlite transformation.
When cooled below the start line for the martensite transformation, austenite will transform into martensite.
Comparison between continuous cooling curve and isothermal C curve
The continuous cooling curve is located to the lower right of the isothermal C curve, with a lower P transformation temperature and a longer duration.
Eutectoid and hypereutectoid steel have a P-transformation termination line, but no B-type transformation during continuous cooling.
For hypoeutectoid steel, subcooling in a specific temperature range during continuous cooling may result in partial transformation into B.
Determining the continuous cooling transformation curve is challenging, so many steels still lack this information.
In practical heat treatment, the continuous cooling transformation process is often estimated by referring to the C curve.
Comparison of TTT curve and CCT curve of eutectoid carbon steel

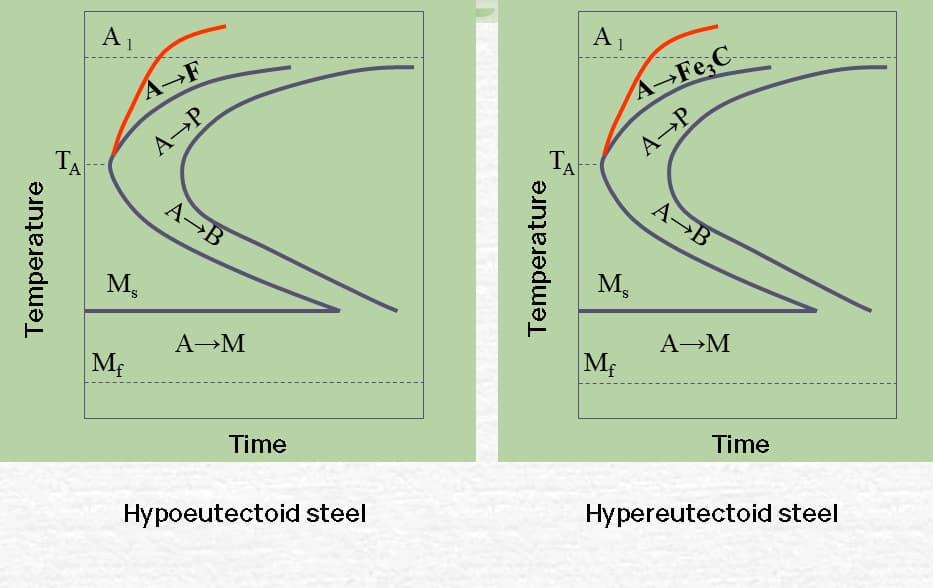
TT curve of hypoeutectoid and hypereutectoid steels
2. Hardenability of steel
(1) Concept of hardenability
The hardness of steel refers to the depth to which the steel can be hardened during quenching, which is a characteristic of the steel.
During quenching, the cooling rate varies at different sections of the workpiece.
The surface cools at the fastest rate, surpassing the critical cooling rate for the formation of martensite. As a result, a martensitic structure is formed after quenching.
As the cooling rate decreases towards the center, if the cooling rate at a certain depth from the surface drops below the critical cooling rate required for the formation of martensite in the steel, then the workpiece will not fully harden as there will be a non-martensitic structure present after quenching.

(2) Effect of hardenability on mechanical properties
The mechanical properties of steels with good hardenability are uniform throughout the section, whereas those of steels with poor hardenability vary along the section. The mechanical properties, particularly toughness, decrease as you move closer to the center.

Fig. 5-53 comparison of mechanical properties of steels with different hardenability after quenching and tempering treatment
a) Hardened shaft
b) Unhardened shaft
(3) Determination and expression of hardenability
There are several methods to determine hardenability. The most widely used method, as specified in GB225, is the end quench test for structural steel. This test measures the thickness of the hardenable layer.
Another commonly used measure of hardenability is the critical diameter. This value represents the maximum diameter of the semi-martensitic structure (50%) that can be achieved at the center of the steel after quenching in a cooling medium. It is denoted as Do.
Top quenching method

Critical quenching diameter



