Have you ever wondered how to select the right welding materials for stainless steel? This article delves into the complexities of welding different types of stainless steel, highlighting key considerations such as matching base materials, dealing with dissimilar metals, and preventing issues like intergranular corrosion. By the end, you’ll have a clear understanding of how to choose the appropriate welding consumables to ensure strong, durable welds. Dive in to learn essential tips that can save you time and enhance your welding projects.

There are four types of stainless steel: austenitic, martensitic, ferritic, and duplex stainless steel (as shown in Table 1).
Table 1 Stainless steel types and their Cr and Ni element contents
| Types | Cr/% | Ni/% | Grade |
|---|---|---|---|
| Austenite | 16-30 | 8-40 | 200,300 |
| Martensite | 11-18 | 0-5 | 403,410,416,420 |
| Ferrite | 11-30 | 0-4 | 405,409,430,422,446 |
| Duplex | 18-28 | 4-8 | 2205 |
This is based on the metallographic structure of stainless steel at room temperature. Upon heating mild steel to 1550°F, its structure transforms from a ferrite phase to an austenite phase. Upon cooling, the structure of the low carbon steel reverts back to ferrite.
The austenite structure at high temperatures is non-magnetic and has lower strength but better toughness compared to the ferrite structure at room temperature.
If the chromium (Cr) content in the steel is greater than 16%, the ferrite structure at room temperature is stabilized, causing the steel to remain in the ferrite state across all temperature ranges. This type of steel is referred to as ferritic stainless steel.
If the Cr content is greater than 17% and the nickel (Ni) content is greater than 7%, the austenite phase is stabilized, allowing the steel to remain in the austenite state from low temperatures to near its melting point. This type of steel is referred to as austenitic stainless steel and is usually referred to as “Cr-Ni” type. Martensitic and ferritic stainless steels are referred to as “Cr” type.
Elements in stainless steel and filler metals can be classified as either austenite-forming or ferrite-forming. The most important austenite-forming elements are Ni, carbon (C), manganese (Mn), and nitrogen (N), while the key ferrite-forming elements are Cr, silicon (Si), molybdenum (Mo), and niobium (Nb). The element content can be adjusted to control the ferrite content in the weld.
Austenitic stainless steel is easier to weld and has better welding quality compared to stainless steel with less than 5% Ni. The welded joints of austenitic stainless steel have good strength and toughness and typically do not require pre-heating or post-weld heat treatment.
In the field of stainless steel welding, austenitic stainless steel accounts for 80% of the total amount of stainless steel used, so this article will focus on welding austenitic stainless steel.
When welding the same base material, it is important to match the base material with the welding material. For example, when welding 310 or 316 stainless steel, you should use the corresponding welding material.
In the case of dissimilar materials, it is recommended to choose a base metal with a high content of alloying elements. For example, when welding 304 and 316 stainless steel, you should choose 316 type welding consumables.
However, there are exceptions to the principle of matching the base material. In such cases, it is important to consult a welding material selection table. For instance, although type 304 stainless steel is a common base material, there is no 304 electrode available.
When welding 304 stainless steel, it is recommended to use type 308 welding material as the additional elements in 308 stainless steel can effectively stabilize the weld area. 308L is also an acceptable alternative. The “L” in 308L stands for low carbon content, with a carbon content of 0.03% or less. In comparison, standard 308 stainless steel can contain up to 0.08% carbon.
L-type welding materials, such as 308L, belong to the same type as non-L-type welding materials but have the advantage of a lower carbon content, reducing the risk of intergranular corrosion (Figure 1).
It is believed that the use of L-type welding consumables will increase as manufacturers aim to improve the quality of their products.

Figure 1 Use of L-shaped welding materials can reduce the tendency of intergranular corrosion
Manufacturers who use the GMAW welding method can consider using 3XXSi type welding materials, as the addition of silicon (Si) can improve wettability (Figure 2).
In situations where the weldment has a high bulge or the weld pool is poorly connected at the toe of the fillet or lap weld, using Si-containing gas shielded welding wire can improve wettability and increase the deposition rate.

In GMAW welding, to improve the wettability of the welding material, a Si-containing welding wire such as 308L Si or 316L Si can be used (Figure 2).
When considering carbide precipitation, a 347 type welding material with a small amount of niobium (Nb) can be chosen as a solution.
To reduce costs, some structural parts may have a corrosion-resistant layer added to their surface by welding carbon steel.
When welding base alloys with no alloying elements and base alloys with alloying elements, a welding alloy with a higher alloy content is used to balance the dilution rate in the weld.
When welding carbon steel with 304 or 316 stainless steel, as well as other dissimilar stainless steels (Table 2), 309L welding consumables are commonly used. If a higher chromium (Cr) content is desired, then type 312 is used.
Table 2 Stainless steels with high alloy content of 309L and 312 are suitable for welding stainless steel and carbon steel
| Ni | Si | C | Mn | Cr | FN WRC-92 | N | Mo | |
|---|---|---|---|---|---|---|---|---|
| 309L | 13.4 | 0.4 | 0.02 | 1.8 | 23.2 | 10 | 0.05 | 0.1 |
| 312 | 8.8 | 0.4 | 0.1 | 1.6 | 30.7 |
It is important to note that the thermal expansion rate of austenitic stainless steel is 50% higher than that of carbon steel.
During welding, the difference in thermal expansion rate can result in internal stress and cause cracking.
To mitigate this issue, it is necessary to select the appropriate welding material or specify the appropriate welding process (Figure 3).
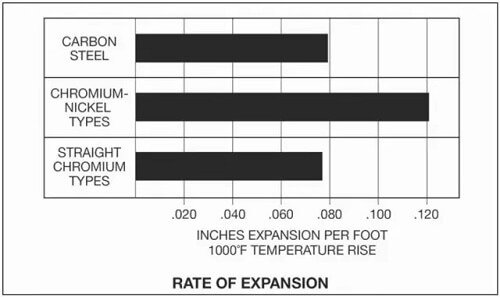
Figure 3 highlights the need for greater compensation when welding carbon steel and stainless steel due to warpage deformation caused by their different thermal expansion rates.
When welding to other materials, it is crucial to first clean the area using a chloride-free solvent to remove oil, marks, and dust. One of the key considerations when welding stainless steel is to prevent contamination by carbon steel, which can compromise the corrosion resistance. To avoid cross-contamination, some companies store stainless steel and carbon steel separately.
When cleaning the area around the groove, use a special sanding wheel and brush specifically designed for stainless steel. In some cases, secondary cleaning of the joint may be required. As electrode compensation is more challenging when welding stainless steel compared to carbon steel, proper joint cleaning is crucial.
To begin, it’s worth noting that stainless steel does not rust due to the protective oxide layer formed by the reaction between chromium (Cr) and oxygen (O).
However, stainless steel can rust as a result of carbide precipitation and heating during the welding process, leading to the formation of iron oxides on the surface of the weldment. Additionally, a seemingly perfect weldment can result in undercuts in rusty areas at the boundaries of the welding heat affected zone within 24 hours.
To regenerate new chromium oxides and prevent rusting, it is necessary to polish, pickle, sand, or scrub the stainless steel after welding. It’s important to note that the sandpaper and brush used must be specific to stainless steel.
Austenitic stainless steel is non-magnetic in nature. However, the elevated temperatures during welding can cause the growth of grains in the structure, leading to an increase in crack sensitivity after welding.
To mitigate hot crack susceptibility, welding material manufacturers add ferrite-forming elements to the welding material (Figure 4). The presence of ferrite phase helps to refine the austenite grains, thereby increasing crack resistance.

Figure 4 illustrates the use of ferrite to prevent hot cracking in austenitic welding materials. Most austenitic welding materials contain a small amount of ferrite, as seen in the picture of the 309L welding consumable, where the ferrite phase (gray part) is distributed throughout the austenite matrix.
Austenitic weld metal is not attracted by a magnet, but a slight suction force is felt when a magnet is held near it. However, this has led some users to mistakenly believe that the product was mislabeled or the wrong welding material was used, particularly when the label on the package is missing.
The amount of ferrite in the consumable is dependent on the service temperature of the application. For example, an excessive amount of ferrite can reduce toughness at low temperatures. This is why the ferrite number of type 308 welding materials used for LNG pipelines is between 3-6, while the ferrite number of standard 308 type welding materials is 8.
In conclusion, while the consumables may appear similar, small differences in composition can have a significant impact.
Typically, the duplex stainless steel structure is composed of approximately 50% austenite phase and 50% ferrite phase. The ferrite phase contributes to improved strength and stress corrosion resistance, while the austenite phase enhances toughness. The combination of these two phases results in even better performance for duplex stainless steel (Figure 5).
The range of duplex stainless steel is quite broad, with 2205 being the most common type. 2205 contains 22% chromium, 5% nickel, 3% molybdenum, and 0.15% nitrogen.

Figure 5 Duplex stainless steel combines the advantages of ferrite and austenite.
The picture shows the dual-phase weld structure of the austenite phase (white part) distributed in the ferrite matrix. However, excessive amounts of ferrite can pose challenges when welding duplex stainless steels, as the heat from the arc can cause atoms in the ferrite matrix to reorder.
To address this issue, welding consumables need to provide more austenite-forming elements, which is usually 2-4% higher nickel content than the base metal. For example, the flux-cored wire used when welding 2205 stainless steel contains 8.85% nickel. After welding, the ferrite content in the weld is typically between 25-55% (and can be even higher).
It is important to note that the cooling rate after welding must be slow enough to allow for reformation of austenite, but not too slow, as this can result in the precipitation of intermetallic phase. Similarly, cooling too quickly can result in excessive ferrite in the heat affected zone.
To ensure the best results, always follow the welding procedure and welding material selection manual provided by the manufacturer.
At temperatures between 800-1600 °F, if the carbon content exceeds 0.02%, carbon (C) will diffuse and migrate to the austenite grain boundaries and react with chromium (Cr) to form chromium carbides.
If too much chromium is fixed by carbon, the corrosion resistance will decrease, leading to intergranular corrosion if exposed to a corrosive environment. This corrosion will result in erosion at the grain boundaries (Figure 6).
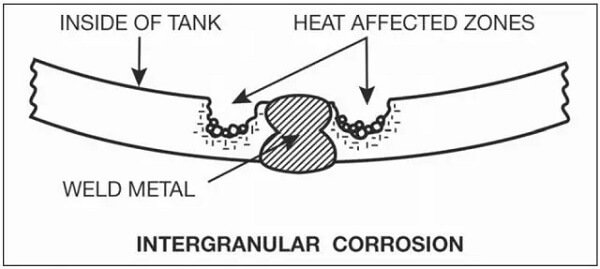
Figure 6 illustrates intergranular corrosion that occurred in the welding heat affected zone of a water tank filled with corrosive medium. To reduce the likelihood of carbide precipitation and improve corrosion resistance, low-carbon or specially alloyed welding materials can be used.
To control carbide precipitation, a low-carbon content welding material is employed to ensure that the carbon content in the weld metal is as low as possible, up to 0.04%. Additionally, the addition of Nb and Ti elements can also fix carbon, as these elements have a higher affinity for carbon than chromium. Type 347 consumables are specifically designed for this purpose.
To select the appropriate stainless steel welding material, it is important to gather information about the end application of the weldment. This includes details about the service environment, such as the service temperature, presence of a corrosive medium, and desired level of corrosion resistance, as well as the expected service life.
Information about the required mechanical properties under service conditions, such as strength, toughness, plasticity, and fatigue properties, is also important.
Most leading welding material manufacturers provide instruction manuals for selecting the materials. It is highly recommended to refer to these manuals or to consult with the manufacturer’s technical experts for help in choosing the correct welding material. This will ensure that the correct material is selected for the specific application and requirements.